
Michael Wildenstein and
Sang Shin
RO. Box 624, Brookfield, WI 53008
WHITE LINE disease has been debated for many years and has been a source of contention between veterinarians and farriers.
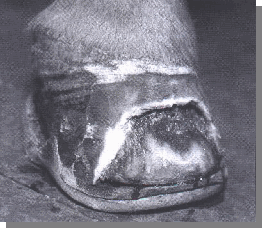
The disease creates a soft, chalky horn tissue in the stratum medium. In severe cases, the stratum internum may be affected. The abnormal horn can be limited to the distal centimeter of the wall, but often extends proximally and can be as high as the coronary area.
The term "seedy toe" has been used to describe this disease due to the "hollowness" of the affected tissue when percussed. Given the fact that the "white line" is technically the junction between the stratum internum and the horn of the sole, the term "white line disease" is probably inappropriate in most cases.
In advanced cases, the white line can be involved, but the majority especially in the early stages are primarily restricted to the stratum medium. The inner-most zone of the stratum medium can be unpigmented and appear white when viewed from the solar surface.
Previous Studies
The horse health literature and farrier trade journals contain a plethora of anecdotal reports and uncontrolled studies on the disease postulating a great number of causes and predisposing factors. A recent summary of this literature (1) discussed some of the suspected predisposing factors, including excessive moisture/drying; lamellar damage (laminitis) acute trauma/ abscess development; hoof cracks; and excessive toe length.
A great number of bacteria and fungi have been incriminated in the development of the disease, but sample collection techniques are either unreported or can be criticized regarding contamination of the samples. In one of the most complete studies included in this White Line Disease, Onychomycosis update by Lubbock, Texas, farrier Burney Chapman, there were 25 different species of fungi incriminated in affected feet. Pseudallescheria sp and Scopulariopsis were two of the more common fungal isolates.
One veterinary report (2) that evaluated tissue under both a regular microscope and an electron microscope, showed white line disease is caused by a fungus.
The questions remain: Is this disease a primary or secondary infection with bacteria, fungi or both? If it is secondary, what are the predisposing factors.?
Attempt To End Controversy
A prospective clinical study currently underway at Cornell University hypothesizes that the term onychomycosis. as offered by Burney Chapman in much of the existing literature, is appropriate. Chapman defines the disease as a fungal infection of the hoof wall secondary to mechanical stresses related to poor management or other disease processes.
All horses presented to our Cornell University tamer service were evaluated for the clinical signs of white line disease. If feet were identified with an irregular white line area consisting of thickening. an abnormal percussion up the wall and a dry, chalky texture. and consent was granted, the horse was selected for the study.
A total of five feet to date have been evaluated with an average age of 5 years (ages ranged from 1 to 8 years). The feet were from four geldings and one mare. Clinical signs at presentation consisted of the aforementioned horn quality in the white line area and variable horn cracking, lameness and dorsal hoof wall separation.
Four horses were selected as the control group because they were determined to have completely normal feet.
A Sample Collection
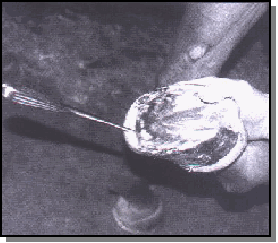
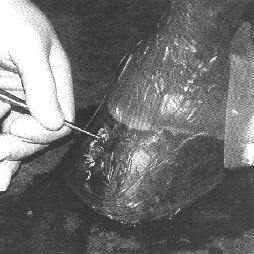
An attempt was made to determine where the leading edge of the lesion was located by using hoof wall percussion. In all suspected cases, there was a distinct hollow sound to percussion that disappeared half the distance or greater from the sole to the coronary hand.
This junction was assumed to be the lesion's leading edge and was chosen for sample collection. An attempt was made to sterilize the hoof wall prior to sample collection.
The hoof wall was painted with concentrated formaldehyde three times at 5-minute intervals. A 1-centimeter-diameter portion of the stratum externum was then removed with a sterilized Dremel bit, and the formaldehyde application was repeated with a final swabbing of concentrated iodine placed in the created wall defect.
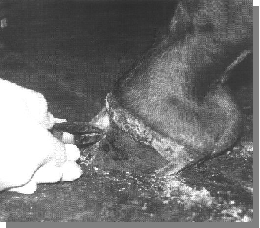
The hoof wall and pastern were covered with an iodine-impregnated sticky surgical drape and a small window was cut out over the created wall defect. Sterile surgical gloves were worn.
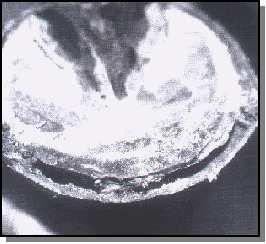
At this point, sterile bone curettes were used to remove the first few millimeters of the stratum medium. which was discarded. Approximately 50 percent to 70 percent of the stratum medium was removed from the 1-centimeter-diameter hole.
The stratum medium collected for culture was dry. chalky and not considered to be of normal texture. This material was transferred into a sterile container for culture.
All samples were aseptically submitted to the New York State Diagnostic Laboratory for aerobic/anaerobic bacterial and fungal culture.
Preliminary Results
The four control feet did not grow bacteria or fungi. The five affected feet did not grow bacteria on either aerobic or anaerobic cultures, but all were positive for the presence of a fungus in the deeper tissues of the stratum medium.
The individual species of isolated fungi were variable and all are considered to be common environmental contaminants.
None of these fungi are known to have the primary ability to cause disease,
although the species Gliocladium is well-known for its ability to cause disease
in plants, specifically roots and tree bark.
The individual fungal species isolated are as follows: Trichoderma sp, Mucor
sp, Aspergillus glaucus and Gliocadium sp (in two feet).
Preliminary Conclusions
The fact that all of our controls were negative for both bacterial and fungal growth validates our sample acquisition as a methodology for collecting sterile samples of the stratum medium for culture. We feel this is a weak link in many of the other studies reporting the presence of fungus in hoof biopsy material. In much of the literature. the sample techniques are either inadequate to ensure against contamination or were not stated to allow for evaluation of the technique. In addition, many of the previously reported studies didn't include control hooves or failed to include bacterial cultures.
The absence of bacteria and positive growth of fungi supports the role of a fungal infection and. therefore, supports the term "onychomycosis" in describing this disease.
An interesting fact is that all four species of cultured fungi are known to potentially be "kerannophilie" (literally meaning keratin "loving"). This does not mean they have the ability to cause primary keratin disease, but have the ability to grow well on keratin.
We continue to hypothesize that mechanical stress created by excessive toe length. laminitis. poor management and/or generally poor hoof health predisposes the hoof to the collection of environmental contaminants within the stratum medium. All identified fungi are envtronmental organisms and can be cultured from soil and wood.
The inner environment of the wall horn (chemical composition, oxygen tension and the limited availability of microbial substrates) selects for species of fungi that can survive and grow well on keratin. It's this collection of factors that likely leads to this condition.
Removal of the affected tissue, sterilization of the underlying tissue and protection/support with an appropriate hoof repair material and shoeing appears to be the most effective treatment.
There are numerous topical products reported to have varying degrees of efficacy. including iodine, bleach, formaldehyde, copper sulfate, DMSO. metronidazole, pine tar, Sav-A-Hoof, Thrush Buster, turpentine, gasoline, merthiolate, benzoyl peroxide, FUNGIDYE, miconazole, clotrimazole, CleanTrax and many others.
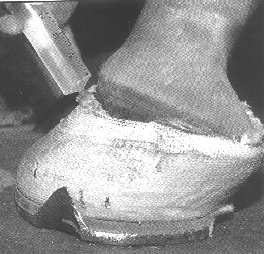
Treatment Continued
The findings of our studies to date support the use of anti-fungal treatments with the development of antifungal-impregnated hoof repair materials being of great interest. It must be mentioned that the antibiotic "metronidazole" is most useful in anaerobic bacterial infections and has little activity against fungi. The bleach products, DMS0, FUNGIDYE and other more classic antifungal drugs, such as miconazole, clotrimazole and itraconazole, should be of benefit.
A Review Of Foot Structure
The wall of the hoof grows from the epithelium covering the coronary dermis, which consists of horn tubules embedded in the intertubular horn that is attached to The coffin bone and hoof cartilages.
The three basic layers of the wall structure consist of the stratum externum, medium and internum. The stratum externum consists of horn tissue produced by the perioplic dermis, which lies directly proximal to the coronary dermis, The stratum externum is only a few millimeters thick, is somewhat rubbery near the coronary band and Is dehydrated over the distal hoof wall.
The bulk of the hoof wall is made up of the stratum medium's pigmented horn tubules. The non-pigmented stratum internum consists of the approximately 600 laminae that interdigitate with the sensitive laminae of the laminar dermis.
The dermis of the sole is firmly attached to The undersurface of the coffin bone and produces a mixture of horn tubules and intertubular horn. The junction between The sole and wall is The so-called "white line" (zona alba). The whIte line includes some of The non-pigrmented stratum medium of The wall,The distal ends of the horny laminae and, befween These, pigmented horn produced over The terminal papillae of The laminal dermis.
References
1. American Farriers Journal Management Report #6: White Line Disease,
Onychomycosis Update. Lessiter Publications, P.O. Box 624 Brookfield, WI
53008-0624. Phone (800) 645-8455.
2. Kuwano, et al. Pathomorphological Findings in a case of Onychomycosis
of A Racehorse, Japan Racing Association. Journal of Veterinary Medical Science,
1996; 58(11):1,117-1,120.
The authors work at Cornell University in Ithica, N.Y. Dr. Michael A. Ball is from the Department of Pharmacology and Large Animal Medicine; Michael Wildenstein is with the Department of Farrier Science; and Sang Shin is with the New York State Diagnostic Laboratory. The Authors would like to thank Steve Bloom of Across The Anvil East, Inc. and Bob Peacock, Farrier Science Clinic, for their financial support of these studies.